Founded in 1993, MEDIANA Co., Ltd. is making efforts to develop products
that can contribute human society based on medical technologies that
measure vital signs and revitalize heart function.
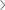 COMPANY
COMPANY
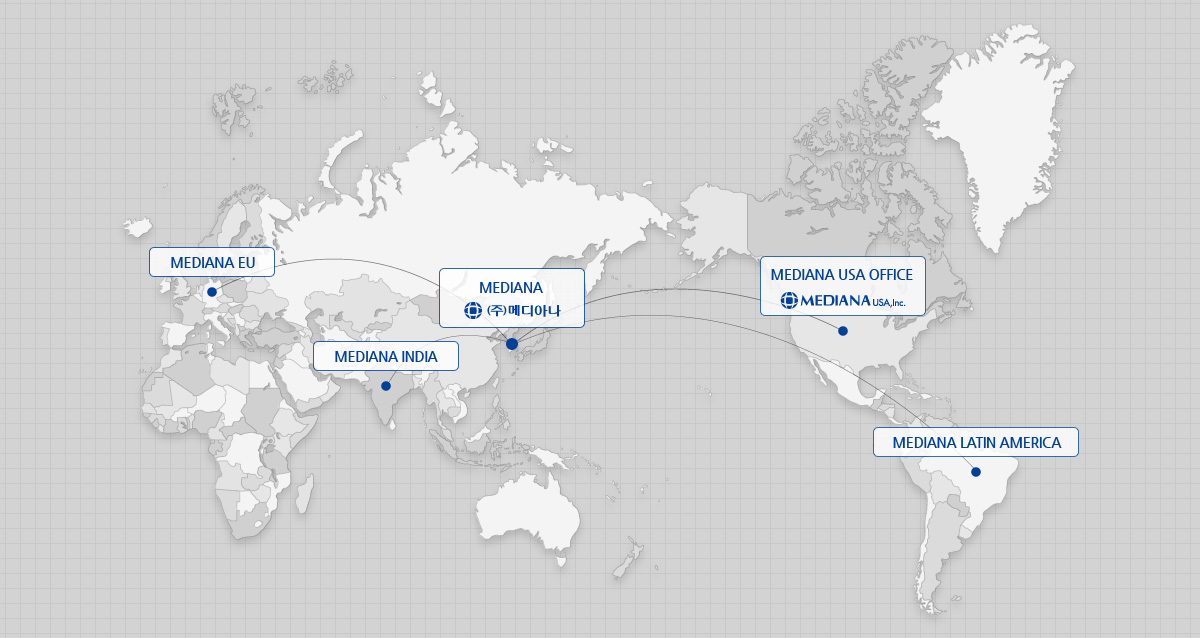
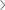 GLOBAL NETWORK
GLOBAL NETWORK
Mediana is a specialized manufacturer of medical devices and is exporting ODM and also our own branded products to all over
the world.
Mediana’s agencies are located in over 80 countries around the world. Over 79% of our total revenue is generated
from exports.
In addition to Mediana, which serves as the head office of operations, subsidiaries are present in the USA, Germany,
India and Brazil..








Obtained from DNV(Det Norske Veritas) in June2000
International certificate on quality guaranty relating to design, development,production, installment and calue-added services.
a quality management certificate that assures safety to European consumers and environmental protection
a quality management certificate of Canada(NSAI)
FDA 510(k) cleared GMP(Good Manufacturing Practice) compliance
CB Scheme, established by the International Electrotechnical Committee for Conformity Testing to Standards for Electrical Equipment (IECEE),
provides a means for the mutual acceptance of test reports among participating safety certification organizations.
UL certification derives from Underwriter’s Laboratory, an organization that tests and certifies electrical devices and appliances for consumer safety.
UL is recognized by Nationally Recognized Testing Laboratory(NRTL), which determines that specific equipment and materials ("products") meet
consensus-based standards of safety to provide the assurance, required by OSHA in U.S.